In this clip we investigate atmospheric pressure. We use a coke can and large petroleum can to show the force that the atmosphere exerts pressure on everything.
Methodology
Materials
- Water
- Coke can
- Petroleum can
- Bunsen burner/Blow torch/Hotplate
Method
Open the coke can and empty the contents. Add 20 ml water to the can. Boil the water in the can. The steam drives out the air in the can creating a vacuum. Put the can into water, the steam condenses leaving a vacuum. The atmospheric pressure then crushes then can. Repeat and upscale the experiment using the bigger can.
Tips
To speed up process use boiled water. Add ice and salt to water to decrease the temperature of water bath.
*Precautions/Safety*
Theory behind the hook
This hook shows the strength of atmospheric pressure. Atmospheric pressure is the force exerted onto the surface of the earth from the column of air above it. This is an invisible force due to the fact that we cannot see or feel it. This makes the concept of atmospheric pressure a more abstract and difficult one for pupils to comprehend. To demonstrate that this pressure exists and to show its strength, the hook crushes aluminum cans.
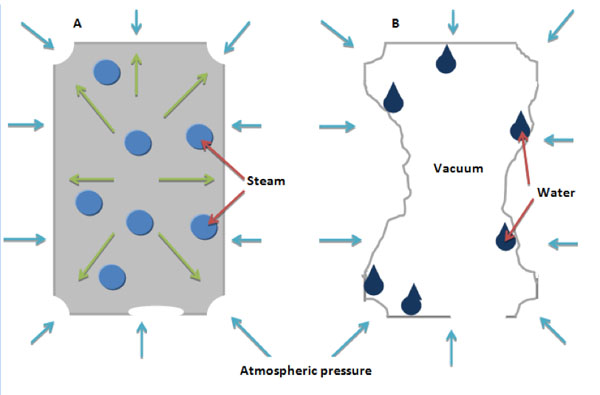
When you boil water in the can, it turns to steam. This steam forces the air out of the can. At this point, the can is full of steam. When you place the can into cold water, the steam in the can loses its heat and reverts back into water i.e. it condenses. Steam takes up a lot more space than water as do all gases over their liquid forms. The water particles in the can take up a lot less area than the steam particles, so when the steam condenses, a vacuum is produced in the can. A vacuum is a region of gaseous pressure that is less than atmospheric pressure. At this point, the pressure outside the can is greater than the pressure inside the can. Atmospheric pressure then crushes the can.
Fig 1. Can A has equal pressure inside and out due to the steam inside the can and retains its shape. In can B the steam is condensed back into water. This leaves a vacuum inside the can and atmospheric pressure crushes it.
How this hook works
This hook is based upon the classic science experiment where cans are crushed using atmospheric pressure. This hook is designed to be used in conjunction with any physics section on pressure. It can also be used on any air section. The hook works on the basis that we cannot feel atmospheric pressure, but it’s a very strong force that is always acting on our bodies. The hook essentially asks the question “do you think that atmospheric pressure is strong enough to crush an aluminium can?” Pupils’ instincts will say “no” and the video uses the element of surprise to gain the pupils attention.
Questions & Answers
Lower Order
- What is atmospheric pressure?
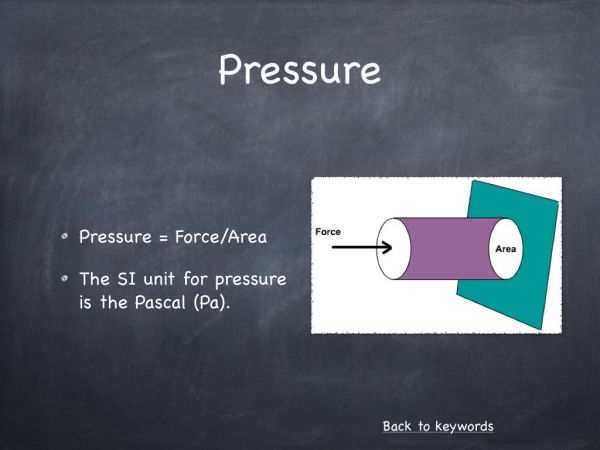
1.01 x 105 Pa - What it the formula for pressure?
Force/Area - What are the units of pressure?
Pascals (Pa) or Newtons per metre
Higher Order
- Why can you not have a proper cup of tea at the top of Everest?
Atmospheric pressure lowers boiling point. When a liquid boils, the particles turn from a liquid form to a gas form. In doing this, the particles gain enough energy to “jump” out of the liquid. At sea level, atmospheric pressure is at its strongest and prevents the particles in a liquid from jumping or moving out of the liquid. This is why the boiling point of water at sea level is 100°C. On top of Mt. Everest, atmospheric pressure is a lot lower. This makes it easier for the particles of a liquid to jump out and become a gas upon boiling. The boiling point of water at the top of Mt. Everest is 72°C. The difference in temperature is why a cup of tea will taste different. - Why is it hard to hold your breath under water?
Pressure due to water. Water exerts a pressure that is similar to atmospheric pressure. The lower you are under water, the higher the pressure is from the amount of water above you. This is why submarines are made from reinforced steel. They need to be strong so that they can absorb the extreme pressure exerted by the water.
Cross Curricular Links
Geography > Weather Systems
Air pressure is not uniform across the Earth. These differences are the result of low and high air pressure systems, which are caused by unequal heating across the Earth’s surface by the sun. Different pressures have different effects on weather patterns. High pressure generally means warm and dry weather, whereas low pressure means cool and wet weather.
Numeracy
Manipulation of the formula Pressure = Force/Area